Shenzhen Creative Industry Co., Ltd was established in 2000. In 2018, it joined Lepu Medical Technology (Beijing) Co., Ltd., the top 2 medical giants among listed companies in China's medical industry. Lepu Medical is a professional medical device manufacturer and company specializing in providing telehealth solutions and focusing on research, development, production, and sales of Telehealth Devices, AI ECG Platform, Smart ECG Machines, Home Medical Equipment, Pulse Oximeters, Holter ECG Monitors, All-in-one Vital Signs Monitors, and Multiparameter Patient Monitors. Our Lepu products have been exported to millions of users all over the world. To date, we are working with business partners in more than 80 countries. We warmly welcome customers at home and abroad to cooperate with us and achieve success together.

Lepu Profile:
Lepu Medical Technology (Beijing) Co., Ltd. was established in 1999. Lepu Medical is specialized in developing, manufacturing, and distributing high-tech medical devices and equipment. Today, Lepu Medical has grown into a global leading group company in the fields of cardiovascular, neurovascular, and peripheral vascular interventions, structural heart diseases, surgical cardiology, cardiac rhythm management, anesthesia, and critical care, in vitro diagnostics and general surgery with products include coronary stents, dilatation balloon catheters, interventional accessories, occlusion devices, mechanical heart valves, electrophysiology catheters, pacemakers, in vitro diagnostic products, critical care products, angiography systems, and surgical staplers.
In 2009, Lepu Medical went public in the ChiNext Shenzhen Stock Exchange market (stock code: 300003). Now Lepu Medical totally has 32 primary subsidiary companies worldwide. Currently, there are 123 products have received CE certificates, and 17 products have got FDA approvals. As the National Interventional Cardiology Medical Instruments & Engineering Technology Research Center, Lepu Medical Group’s top strategy is to build up a 4 in 1 platform that includes medical devices, medicines, health services, and mobile medicals for cardiac treatments.
Lepu Medical has established three overseas subsidiary companies in Netherlands, Turkey, and India obtained local registrations in 27 countries, and built sales and distribution channels in over 80 countries and regions. Lepu Medical upholds “integrity, quality, and scientific innovation” as its spirit and commitment to providing the best quality of products and services to satisfy the needs of healthcare professionals and patients. This catalog includes information on products available in certain countries.
Lepu Vision:
To develop Lepu Medical into one of the largest suppliers of cardiovascular-related products and services on the platforms of medical devices, medicine, healthcare products, healthcare service, and new medical mode.

Creative was established in Shenzhen.

Household products passed the US FDA510(k) registration and approval for marketing.

Excellent information technology innovation enterprise award gold award.

The whole line of products passed the TUV CEO123 certification and passed the TUVI ISO13485 quality system certification.

The oximeter product passed the Japanese MHLW No.169 quality system certification.

The monitor product passed the US FDA 510(k) registration and was approved for marketing.

Joined Lepu Medical Technology (Beijing) Co., Ltd.

Guangdong provincial quality and credit class a medical device manufacturing enterprise.
It will rank 28th on the Shenzhen medical device enterprise billion list.
In terms of production management, the company fully implements the ISO9001:2000 international quality management system, strictly implements the Chinese medical device industry standard YY/T0287—ISO13485, and has passed the quality certification of the China Medical Device Quality Certification Center CMDC, and all products have passed the EU CE certification. Some products have been approved by the FDA of the United States.





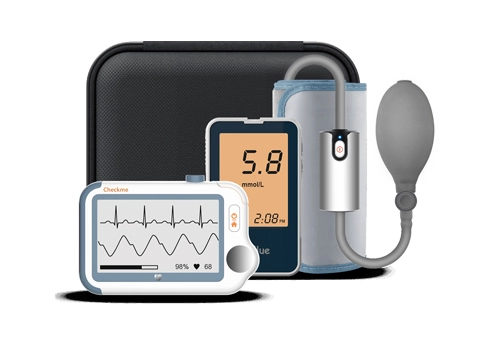
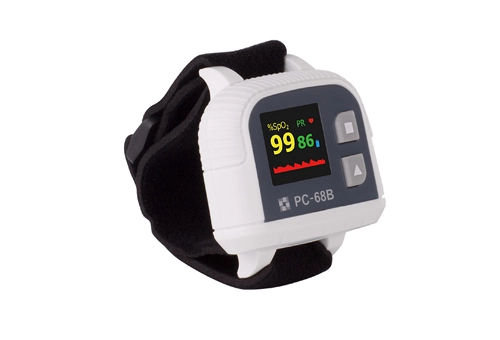
Related Lepu-Creative Telehealth & Medical Monitoring Devices